|
Medical Tubing And Extrusion Technology
Advances
& Limitations
Advances in
Medical polymeric tubing science and extrusion technology
directly correlate to the evolution from surgical-based
intervention to minimally invasive approaches and
associated medical device design. The goal of reducing
procedure and hospitalization times, and patient trauma,
while improving outcomes has created a dependency on
tubing and extrusion science to keep pace with clinical
demands.
Medical
tubing plays a major role in promoting cost efficiency
because cardiac and urinary catheterization that involves
the use of catheter tubing, greatly reduces the time taken
for the concerned medical or surgical procedure. As a
result, the hospital stay is shortened, which, in turn,
lowers the overall charges a patient is likely to pay for
treatment.
Today’s
medical device engineers must understand the advances and
limitations of tubing technology as defined by both the
extrusion process and post-extrusion technology in order
to take advantage of the best designs and ensure the
success of their products. Medical device manufacturers
are being challenged by strong market demand for tubing
that delivers increased functionality, lower profiles, and
lower costs - pushing the limits of material behavior and
manufacturing science. Next-generation balloon catheters
are expected to deliver significantly higher burst
pressures and better puncture resistance. They are also
being designed to transport target-specific drug polymer
payloads or flexible microelectronic packages to various
parts of the human body. Designs are calling for ever
smaller and more capable medical devices, and many are
made to last only one operation. Smaller tubes accommodate
the trend to less invasive procedures because less
traumatic procedures shorten patient recovery times. The
challenge is to make them tight enough for a reliable
design in terms of how the product will be used, yet loose
enough to facilitate assembly and avoid unneeded features
and costs.
Braiding
and coiling can improve performance in medical device
applications by increasing torque force or the bend radius
of a tube. Designing and developing complex multiple lumen
tubing can spur innovation, provided that the designer
understands the latest technology and how best to work
with a tubing manufacturer.
Tubes and
Catheters Sizing
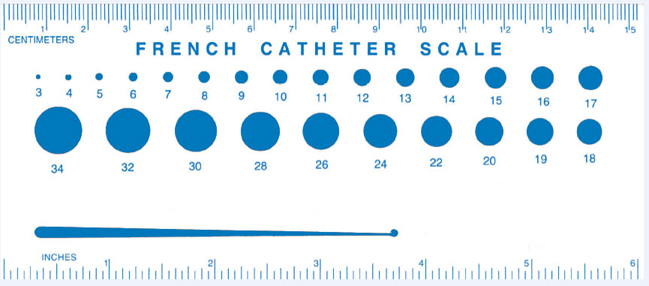
Problems
can arise when the customer and catheter or tubing
supplier are not using the same scale when discussing
catheter tubing sizes.
French
scale and gauge scale are the measures used to refer to
the diameter of catheters and tubing. While each have
certain advantages, misinterpreting these standards can
lead to confusion . “OD” and “ID” are abbreviations for
outer diameter and inner diameter, respectively, and are
generally measured in inches or millimeters.
The
catheter sizing classification most commonly used in the
medical industry is the French (Fr) scale, also known as
the “Charrière’s system.” The French scale is an easy
method to use to describe the basic size of a catheter,
because it specifies the most obvious component, the outer
diameter. The French size = 3 times the diameter in
millimeters. The disadvantage of the French scale is that
it does not specify the inner diameter of the catheter or
tubing. Thus, the French sizing used alone, is not
specific enough to adequately describe small catheters,
where internal diameter is critical. This scale is much
more useful to specify larger catheter sizes.
A second
method of sizing catheters is the gauge measurement. Gauge
is a descending scale, opposite the French scale of
measurement which ascends with corresponding size. The
higher the gauge size the smaller the tubing.
Another
less common method for sizing catheter tubing is the “PE”
scale. It is specific to polyethylene and is an ascending
scale used by some manufacturers to represent the OD and
ID of the tubing, similar to French sizing.
The most
definitive method to describe a catheter’s dimension is
simply to state the exact measurements of both the outer
and inner diameters. While it may seem a little more
difficult to communicate, this method will actually give
all the details necessary to the researcher or catheter
manufacturer to properly size your specific catheter and
catheter connections.
(Ref:
http://www.alnmag.com/articles/2011/08/french-gauge-odid-mm-inches-what-does-it-all-mean
https://en.wikipedia.org/wiki/French_catheter_scale)
Page
1 :
2 :
3 :
4 :
5
|
