|
Simplifying Set-Up of Plastics Clean-Rooms
By : Medline Department, The Conair
Group, USA

Navigating the highly regulated world of medical
manufacturing and clean room operations can be a daunting,
time-consuming task. Regulations and standards, developed
by such organizations as the U.S. Food & Drug
Administration, the International Organization for
Standardization (ISO) and others, are many and complex.
For those who want to begin manufacturing medical plastic
products and components, understanding the regulatory
requirements is only the first hurdle to be overcome. Then
specialized facilities, including cleanrooms (Figure 1),
white rooms and hybrid rooms, need to be designed and
built, and processing equipment needs to be sourced with
special attention not only to performance, efficiency and
quality, but also to cleanliness, calibration, maintenance
and record-keeping. Mistakes can result in delayed
start-up, lost production, quarantined parts,
rework and lack of process validation.
To simplify the process of specifying, buying, installing,
qualifying and operating plastics auxiliary equipment in a
clean environment, Conair has introduced a line of
products – 170 units in all – for molding and extrusion of
medical products (Figure 2). The new MedLine® product line
includes equipment for conveying and storage, resin
drying, blending, heat transfer and downstream extrusion.
All are based on proven Conair designs, but they have been
specially sized, configured, documented and supported for
use in cleanrooms and other controlled environments. Here
is why that’s important:
REGULATORY SITUATION
The regulations that govern today’s cleanrooms were first
developed in the US Federal Standard 209E Airborne
Particulate Cleanliness Classes in Cleanrooms and Clean
Zones. As cleanrooms became more sophisticated, the
industry recognized a need for more wide-ranging standards
for cleanroom classification and testing. This led to the
formation of ISO Technical Committee 209, which was tasked
with the "standardization of equipment, facilities, and
operational methods for cleanrooms and associated
controlled environments." Today, the most common standards
governing “the methods, facilities, and controls” used in
manufacturing, processing, and packing of plastics medical
devices are :
-
ISO 9001 – based on a number of quality
management process approach and continual improvement and
aimed at enhancing customer satisfaction through continual
improvement of systems and processes, and conformity to
customer and regulatory requirements.
-
ISO 14644 - a tiered structure that allows
a specific maximum number of particles per cubic foot of
air cleanliness in various classifications of cleanrooms
and other controlled environments. (Similar to US Fed Std
209E.)
-
ISO 13485 – a quality management system
where an organization needs to demonstrate its ability to
provide medical devices and related services that
consistently meet customer requirements and regulatory
requirements applicable to medical devices and related
services.
-
ISO 17025 – the single most important
standard for calibration and testing
-
FDA 21CFR – current good manufacturing
practices (CGMP) for medical devices and pharmaceutical
packaging.
These standards and regulations provide the checks and
balances that lead to consistent and predictable outcomes
in reduced product variation. Following their guidelines,
and using good cleanroom management techniques, will help
ensure quality and reduce costly mistakes in the
production of medical plastics components.
In developing the MedLine family of equipment, Conair
carefully considered each of these standards. Then they
identified equipment features, calibration standards and
documentation that can help processors get to a
state of compliance faster and more easily, whether they
need a single piece of equipment or a complete system.
DIFFERENT LEVELS OF CLEAN
Depending on the products being manufactured, medical
plastic processors have adopted several different
configurations for clean environments. Each requires a
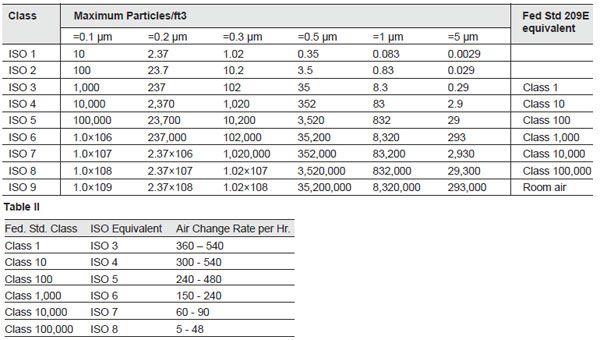
different level of clean. See Tables I and II.
A White Room is a typical entry-level clean environment
meeting ISO 9 standards. Although a white room is
typically air-conditioned, there is no attempt made to
control particulate or air change-over. Otherwise,
it is treated like a cleanroom, with gowning, hairnets and
shoe covers. Typical applications would include lip-balm
tubes, pill holders, medical cases, thermometer housings
and the like. Although standard equipment has been used in
white room environments for many years, there is
increasing focus on the cleanliness and compliance.
Calibration, preventive- maintenance, and installation
qualification procedures are becoming more commonplace.
ISO 8 or Class 100,000 cleanroom
applications – for instance, pumps, auto-injectors,
blood-bag components, diagnostic and testing devices –
require an approach that limits particulate generation by
equipment and personnel working in the room. These rooms
generally have air turnover rates of 5 to 48 times per
hour in order to keep particulate levels in compliance
with the standards. The equipment used in ISO 8
environments must be designed and manufactured to minimize
generation of particulate, chemical vapors and airborne
microbes. ISO 13485, which governs the manufacture of
medical devices, generally requires this or more stringent
levels of cleanliness. Equipment used in an ISO 8
environment usually is made with anodized aluminum,
certain acceptable powder coatings, chemical resistant
HMIs (to allow antiseptic wipe down of surfaces), and even
urethane casters that allow machines to be moved for
cleaning without generating rubber or vinyl particulate.
ISO 7 (Class 10,000) cleanrooms have more
stringent requirements. These rooms generally have air
turnover rates of greater than 60 to 90 times per hour.
Humidity control is an important factor in controlling
static electricity and bacteria growth. Because of this,
many of these rooms also require bio-burden testing (to
measure the number of micro-organisms on surfaces),
antimicrobial- coated materials on room wall panels,
equipment coverings and other surfaces. Room air
ionization also becomes more important. This much-cleaner
environment is much more suitable for pharmaceutical and
sterile device packaging, syringes, vials and implantable
devices. Equipment, then, needs to be designed and
manufactured with consideration for these more stringent
requirements. That means stainless-steel housings,
castings, and covers instead of anodized aluminum, as well
as FDA-complaint gasket materials and plenum-rated
cabling. Particulate control is much more important and so
equipment typically will be cooling-fan-free, using water
cooling or remote exhaust valves. These are just a few
things that need to be considered for an ISO 7
environment.
|
