|
Polymers And Medical Device
Manufacturing
 The
changing technology trends in medical polymers and its
processing techniques contribute immensely towards the
development, growth and innovations not only for Medical
Device Industry but for the new polymeric materials. This is
possible with close interaction between medical polymer
manufacturers and device manufacturers. This is a great
contributing factor for addressing unmet needs in medical
device manufacturing. The
changing technology trends in medical polymers and its
processing techniques contribute immensely towards the
development, growth and innovations not only for Medical
Device Industry but for the new polymeric materials. This is
possible with close interaction between medical polymer
manufacturers and device manufacturers. This is a great
contributing factor for addressing unmet needs in medical
device manufacturing.
The medical device market is booming with
innovation. Medical Device industry needs to stay on top of
emerging trends by successfully blending new materials into
device developments. This is applicable not only for new
products but also to improvements in existing designs.
At the same time, the emerging polymer
technologies for Medical applications must always be seen in
combination with changing and evolving regulatory boundaries.
Emerging Trends In Medical Polymer
Materials & Processing
The following are few of the examples of
important medical device made with the help of developments in
medical polymers & their processing.
PEEK For non-metal lumbar cage in Japan
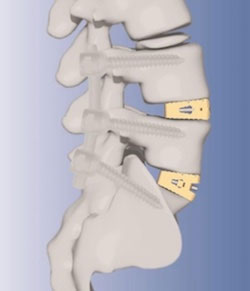 FDA
cleared the first foot-and-ankle implant made from Invibio
Biomaterial Solutions’ PEEK-Optima HA Enhanced polymer comes
word that Solvay’s Zeniva PEEK was selected by medical device
OEM KiSCO Co. (Kobe, Japan) for the first non-metal lumbar
cage for vertical interbody fusion madein Japan. The advanced
spinal cage is commercially available
only in Japan, where that country’s Pharmaceuticals and
Medical Devices Agency (PMDA) approved the implantable medical
device for production. FDA
cleared the first foot-and-ankle implant made from Invibio
Biomaterial Solutions’ PEEK-Optima HA Enhanced polymer comes
word that Solvay’s Zeniva PEEK was selected by medical device
OEM KiSCO Co. (Kobe, Japan) for the first non-metal lumbar
cage for vertical interbody fusion madein Japan. The advanced
spinal cage is commercially available
only in Japan, where that country’s Pharmaceuticals and
Medical Devices Agency (PMDA) approved the implantable medical
device for production.
It is said that the materials was selected
because of its performance properties, including fatigue
resistance and a modulus similar to bone. The company The
wanted an implantable medical material
that could facilitate post-operative diagnosis. PEEK is an
alternative to titanium, which is typically used in this
application, because its radio transparency allows for easy
visualization in medical X-ray and MRI imaging.
The primary function of a spinal cage is to
maintain the proper intervertebral angle and height until
spinal fusion occurs. However, spinal cages also need to
accelerate bone fusion and support adjustments during surgery.
The company’s spinal cages allow surgeons to use a
screw-and-spacer mechanism to make these necessary
adjustments.
During clinical trials, the material
enabled the spinal cage to maintain structural stability even
when screws were used to expand the spacers.
The materials supplier’s ability to provide
quality data for biological evaluation and its responsiveness
facilitated the regulatoryprocess, allowing device
manufacturer to secure approval from PMDA in a short period of
time, said the company.
New Grade Of Silicone Elastomer For
Microfluidic And Implantable Medical Device Applications
Materials innovator Gelest Inc.
(Morrisville, PA) has introduced ExSil 50, a softer grade of
an ultra-high elongation material that has been formulated to
meet the requirements of soft-tissue implants or
extracorporeal device applications.
The ExSil class of elastomers have
unprecedented elongation greater than 5000% and shape recovery
behavior, according to Gelest. They also have self-sealing and
tear-resistance properties. The materials are available in an
industrial grade, GelestExSil 100, as well as soft-tissue
compatible ExSil 50.
Polyarylamide Resin For Single-Use
Bone-Fixation Device
 In
developing a more robust, single-use bone-fixation system than
currently available devices, medtech startup Reign Medical
(Tulsa, OK) went on the hunt for a biocompatible material with
exceptional stiffness properties. A polyarylamide (PARA) resin
from Solvay (Alpharetta, GA) stood out and is used to mold
several components of the medical device soon to be on the
market. In
developing a more robust, single-use bone-fixation system than
currently available devices, medtech startup Reign Medical
(Tulsa, OK) went on the hunt for a biocompatible material with
exceptional stiffness properties. A polyarylamide (PARA) resin
from Solvay (Alpharetta, GA) stood out and is used to mold
several components of the medical device soon to be on the
market.
The Clench compression staple system
comprises a disposable sterile surgical tool set for fixating
hand and foot bone fragments and performing related
procedures. A patented threaded hub allows surgeons to
incrementally expand the implant for proper insertion while
retaining the mechanical properties of the nitinol staples for
continuous compression across the fusion site after
implantation. Reign Medical specified Solvay’s 50%
glass-fiber-reinforced grade, for the device’s implant sizer,
drill guide and each part of the implant delivery instrument,
including its handle, the saddle on which the staple sits and
the threaded compressor that forces the staples fully open,
said Solvay in a press release.
A high-stiffness material was required for
this application because of the mechanical strain that the
components experience. Offering metal-like strength, rigidity
and dimensional stability, the biocompatible polymer has to
provide an exceptional surface finish .Gamma-stabilized colors
are optimized for sterilization.
Importance Of Compounding In Medical
Device Sector
Catheters used for vascular and other
critical care applications need polymeric tubing requiring
high precision, close tolerances and high level of
functionality. This is particularly used for minimally
invasive surgical techniques.
Along with the above mentioned properties,
it is also necessary important to consider manufacturing
process required to manufacture the medical device.
The inside diameter of the catheter is
required to be very smooth to allow the guide wire to slide.
For catheter balloon manufacturing, it is
important that the extruded tubing is amorphous prior to the
balloon forming process.
In order to arrive at the right grade of
compounded material, the device manufacturer has to work with
the material supplier to meet not only the processing /
manufacturing requirements but also the regulatory needs.
|
