|
Gujarat – A Pioneer & No 1
Manufacturing Hub For Life Science Industry In India
|

Dr. H G Koshia,
Commissioner
Food & Drugs Control Administration, Gandhinagar (Gujarat) |
In an extensive interview with “Medical
Plastics Data Service”, Dr H G Koshia shares important
facts & figures regarding the achievements of the state
including Pharmaceuticals, Medical Devices, Diagnostics
and Biotechnology Sub-Sectors. He also shares about the
vital and catalytic role played by Food & Drug Control
Administration (FDCA), Gujarat.
|
Gujarat –A
leading State For Medical Device Manufacturing In India.
With around 300 licenced manufacturers
including high end products.
Manufacturing 78 % In Cardiac Stents,
50% intraocular lenses, 35 % Diagnostic Reagents and large
number of other critical care products. Companies
Recognised Globally.
Gujarat Pharma Industry : No 1 In
India with :
3300 mfg. units, contributes 33 % of India’s Pharma sector
turnover, 28 % of country’s Pharma exports, received 16 % FDI,
Also 40 % of Pharma Machinery Manufacturing in India.
FDCA Gujarat : Pioneer in initiating
Medical Device Regulatory Process in the Country with vital
role in formulating Medical Device Rules.
Played active role to educate, counsel and guide medical
device companies to obtain licenses.
FDCA Gujarat played catalatic role in
promoting Largest Number of COVID 19 Management Products both
in Medical Devices as well as Pharma categories like PPEs.
Hydroxycholoquine and Azithromycine, Medical oxygen, hand
sanitizer, Remdesivir etc. within very short time.
Challenges faced By Guj. Medical
Device Companies :
- multiple government departments wanting to regulate the
industry
- Uncertainty on the policy of price capping
- Needs Better Infrastructure Support
- Taxes
Contribution of Gujarat FDCA in the
Growth of Gujarat Medical Device Industry
-
Before New Medical Devices rules came in existence the devices
were coming under Drugs under Drugs & Cosmetics Act, 1940
rules thereunder.
-
Because of the suggestions and technical inputs from Gujarat
FDCA new medical devices rules were came in existence in 2017.
-
Gujarat played a vital role in the department of Medical
Devices rules.
-
Initially manufacturer manufacturing such kind of devices were
out of regulations. Hence after the new medical devices rules
FDCA played active and important role to educate, counsel and
guide all the manufacturers to obtain license.
-
Hence today there are around 300 licenced manufacturers
including manufacturers of high end products in the
cardiovascular, orthopaedic, ophthalmic and other critical
care categories.
Medical Device Industry Statistics
The Indian Medical Device Industry is
amongst the top 20 markets for Medical Devices in the world
and is 4th in Asia
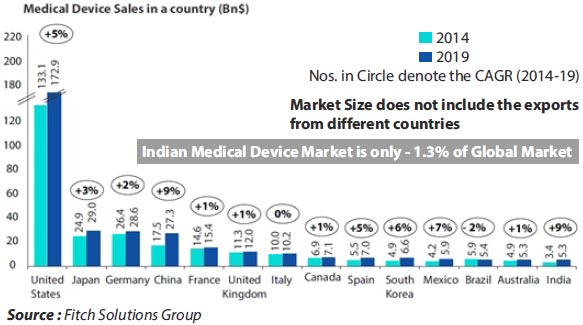
However a significant part of the market
remains import dependant (drive by high end engineering
complexity and high end innovation)
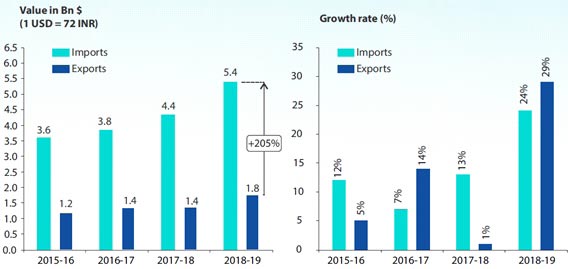
Source : Ministry of Commerce, Industry
Estimates, PwC analysis
Note : Some of the import products are used in exports also,
and hence a higher number for imports than domestic sales
|
