|
In
Vitro Diagnostics, A Key Contributor to Medical
Devices Sector The
world’s eyes are on India and the Government of
India has woken up to the huge potential of growth
offered by the medical equipment and the medical
electronic sector. Ministry of Electronics- Meity
is now treating this as a focus sunrise sector and
has been in consultation with AiMeD and other
stakeholders to replicate the success of the
Cellular Phone industry.
Noteworthy initiatives in this direction include
the establishment of six medical devices
manufacturing “clusters” in the country, including
Andhra Pradesh, Telangana, Tamil Nadu, and Kerala
which are either fully operational or have got in
principle approval. The new
scheme for medical devices and creation of medical
device parks would certainly empower the creation
of a comprehensive healthcare network that can
provide capital intensive facilities required by
devices manufacturers along with contemporary
manufacturing units capable of playing a critical
role in meeting the country’s demand.
Current COVID-19 pandemic has shown that
diagnostics is an essential part of preventive
healthcare in every country. The domestic
diagnostic industry is still at a nascent stage,
estimated at $9 billion (around Rs 675 billion)
and is expected to grow at a CAGR of approximately
15% over the next five years. “Make in India and
the goal for an Aatma Nirbhar Bharat can provide
the required fillip to achieve this growth. To add
to that, the pandemic has further provided the
necessary impetus to scale-up domestic production.
In Vitro Diagnostics (IVD) Sector constitute a
significant part of the healthcare component and
this sector has in recent times witnessed immense
progress largely due to technological
advancements. Cutting-edge technologies have paved
the way for the diagnostic sector to become a
rapidly growing industry in the Indian medical
devices market which can be broadly classified
into equipment, reagents, and services.
Global IVD market is estimated at USD 85 Billion
and India accounts for approximately USD 1.5
Billion (1.8% of global revenues). There has been
a steady increase in the percent contribution of
India to the overall global market over years and
the key drivers include “Health Awareness”,
“Demand for Quality Healthcare”, “Rise in Disease
Prevalence”, “Advances in Clinical Research” and
“Evolution of Newer Technologies”, amongst others.
Government of India is beginning to recognise the
importance of diagnostics in preventing and
treatment of diseases, pointing to the allocation
of Rs 64,000 crores in the budget for FY 2021 22,
with a focus on preventive healthcare and this
should aid in boosting local manufacturing of
diagnostic equipment and test kits in India.
IVD Market in India is hugely dependent on imports
(approximately 75-80% of instruments and reagents
are imported). There has been enormous focus on
reducing the dependence on imports to make India
self-sufficient and can meet the needs of the
market where the imported products and solutions
are not affordable. “Make in India”
and “Atmanirbhar Bharat” are the key
drivers in this direction. It is estimated that
India has approximately 100,000 laboratories (70%
IVD and 30% Radiology) and is
anticipated to grow at 15-18% per annum. It
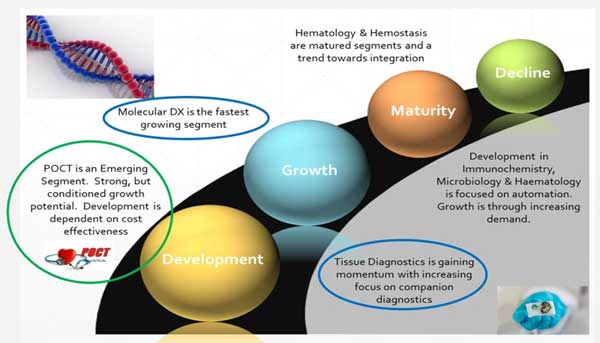
is essential to consider two key aspects when we
discuss the development of clinical diagnostic
segment. This includes the consideration of
“Evolution of IVD Market” and the second being
“Evolution of IVD Technologies”.
Medical device companies are transforming
themselves from devices / consumables providers to
disease / care management organizations to address
the challenges of
accessibility and affordability (more specifically
the rural markets). Connected devices utilizing
digital platforms are making strong in-roads in
this direction. |
