|
Medical Device
Manufacturing Machinery / Equipment

Life-enhancing materials for Healthcare
Patients depend on doctors. Doctors depend on medical
devices and “Those who design medical devices depend
on Covestro”, one of the leading producers of high
performance plastics around the world.
Covestro medical grades meets the below global
regulatory requirements
Biocompatibility: ISO 10993-1 and USP Class VI for
contact of 30 days or less.
Compliance to Regulation (EU) 2017/745 on medical
devices and (EU) No. 722/2012 regarding medical
devices manufactured utilizing tissues of animal
origin.
Maintenance of FDA Device (MAF) and Drug Master (DMF)
File and provide letters of authorization.
Product stewardship.
Manufactured at ISO 9001 certified sites that follow
GMP standards.
Supplier notification of change.
Polycarbonates
PC, PC+ABS, PC+Polyester, High temperature PC
Applications: Insulin pens & pumps, inhalers, renal
care and blood management to heart defibrillators,
intravenous access components and surgical
instruments.

Thermoplastics Polyurethane
Bridges the gap between rubber and hard thermoplastics
Sterilizable
Prevents tube blockages
Excellent resistance to chemicals, lipids, cleaning
agents and bacteria
High Moisture Vapor Transmission Rate for comfortable
wearables and breathable wound dressing.
Applications: Flexible tubing, catheters, connectors,
film and fabric coatings, component housings,
healthcare, wearables.
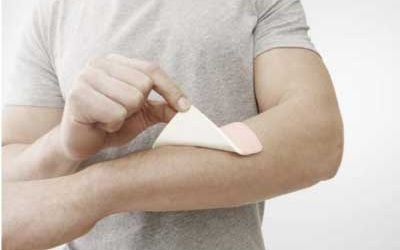
Functional film
TPU films for Wound Care, Surgical (Body Drapes and
Robotic Arm Covers) Theraphies, Diagnostics and
Wearables.
Customised films for Advanced wound care.
Consistency, quality, reliable supply, global product
availability, innovative materials meeting rigorous
healthcare requirements, excellent technical and
regulatory service is what makes Covestro a partner of
choice.
India (covestro.in)
Contact : email-
suresh.v@covestro.com
Mob : +91 9840853179 |
