|
Advances In Asepsis
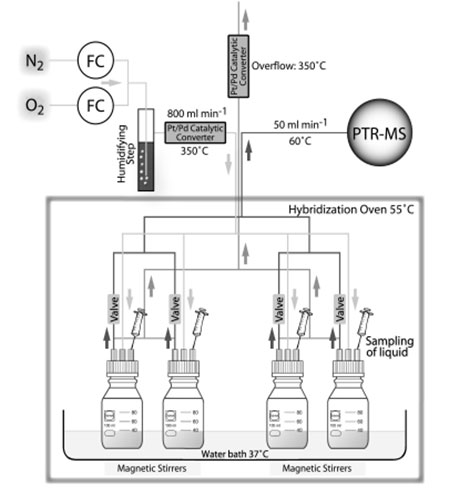
On-linemonitoring of
microbial volatile metabolites by proton reaction mass
spectrometry
Risk Management
Integrate risk management processes across entire
quality management system. This ensures that
nonconformities, regardless of source, are evaluated
using consistent criteria to determine risk using
standards such as ISO 14971
ISO
14971 defines the international requirements of
risk management systems for medical devices, defining
best practices throughout the entire life cycle of a
device.
What are the benefits
of ISO 14971?
-
Implement ideal methods
of reducing risk for all stakeholders
-
Develop devices and
therapies that are proven effective in the industry
-
Manage speed and cost to
market
-
Optimize speed of
iteration
-
Streamline the
regulatory process that will enable entry to
selected markets
|
|
ISO 11607-1:2006 specifies the requirements
and test methods for materials, preformed sterile
barrier systems, sterile barrier systems and
packaging systems that are intended to maintain
sterility of terminally sterilized medical devices
until the point of use. |
A sterile barrier system is an
essential part of a sterile medical device. (A
sterile barrier system is defined as “the minimum
packaging configuration that provides a microbial
barrier and allows aseptic presentation of the product
unit at the point of use”
This is distinct from protective
packaging which is defined as the “packaging
configuration designed to prevent damage to the
sterile barrier system and its contents from the time
of their assembly until the point of use”.)
Quality Management System
The primary objective of ISO
13485:2003 is to facilitate harmonized medical
device regulatory requirements for quality management
systems.
As a result, it includes some
particular requirements for medical devices
Excludes some of the requirements
of ISO 9001 that are not appropriate as
regulatory requirements.
Because of these exclusions,
organizations whose quality management systems conform
to this International Standard cannot claim conformity
to ISO 9001 unless their quality management
systems conform to all the requirements of ISO 9001.
All requirements of ISO
13485:2003 are specific to organizations providing
medical devices, regardless of the type or size of the
organization.
Biomedical production of
implants by additive electro-chemical and physical
processes
Electrospinning
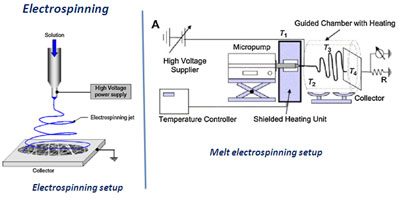
From:
Bartolo P, Kruth Jean-Pierre, Silva J, Levy G, Malshe
A, Rajurkar K, Mitsuishi M , Ciurana J , Leu M.
Biomedical production of implants by additive
electro-chemical and physical processes, CIRP Annals -
Manufacturing Technology 61 (2012) 635–655
|
