|
Promotion of Domestic Production of High End Medical
Devices
The
Government is going to set up Medical Devices Parks in the
country, and first such park will come up in Gujarat soon.
The Union Minister of Chemicals and fertilizers Shri
Ananth Kumar said this while releasing the recommendations
of the Task Force on Promotion of Domestic Production of
High End Medical Devices & Pharmaceutical Manufacturing
Equipment. He said that only 30% of the country’s
requirement of Medical Devices is being met through
indigenous production, and that of low end category. The
Minister said that the Government is taking several steps
to boost the domestic production of high end medical
devices & pharmaceutical manufacturing equipment. He said
that NIPER, Ahmedabad will be the nodal institute for
undertaking research and development in the area. Shri
Ananth Kumar said that the recommendations made by the
Task force will be implemented in a time bound manner. He
said that the Department has already taken up issues of
incentives and taxation on such devices with the finance
Ministry so that the indigenously produced medical devices
can compete with the imported ones. The minister said that
need is being felt to have a separate department to handle
the issues of facilitation and regulation of high end
medical devices & pharmaceutical manufacturing equipment,
as their consumption is increasing very fast and there is
need to make India self-dependent in the sector. He said
that on the lines of National List of Essential Medicines,
there should be a National List of Essential Medical
Devices to enable providing affordable, quality products
to masses easily.
The
Minister of state for chemicals and fertilizers Shri
Gangaram Hansraj Ahir said on the occasion that the
Government is working hard to achieve the objective of
“Make in India”.
Medical
devices’ industry is a multi-product industry, producing
wide range of products. Taking all the products category
of
‘Medical
Devices’ together, India is importing around 69% of its
total requirement. The imports of high-end Medical devices
are much higher, going up to around 87% for the category
of Medical Electronics, Hospital Equipment, Surgical
Instruments. In view of the prevailing scenario, the Task
Force on ‘Promotion of Domestic Production of High - end
Medical Devices and Pharmaceutical Manufacturing
Equipment’ was set up by the Department of Pharmaceuticals
as an initiative to implement the PM’s visions of ‘Make in
India’. The Task Force was headed by Secretary ,
Department of Pharmaceuticals and consisted of
representatives from Planning Commission , Department of
Health & Family Welfare, Department of Health Research, DCGI, Department of Commerce, Department of Industrial
Policy and Promotion, Department of Electronics and
Information Technology and various Industry Associations
including AIMED, CII, FICCI, ASSOCHAM, FOPE etc.
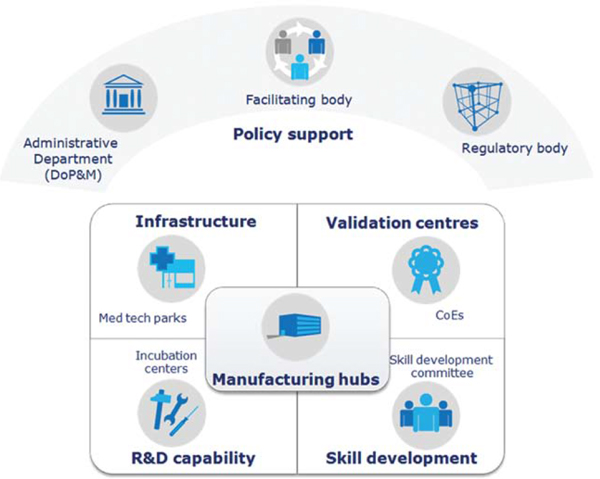
The Task
Force has given recommendations to address concerns and
constraints facing the sector and suggested measures that
would augment domestic production of medical devices. The
Task Force has recommended measures to enhance policy and
institutional support; suggested strengthening of
infrastructural support; and, recommended fiscal and
financial support to the sector. In addition, the Task
Force has also recommended measures for creating an
institutional set-up for efficacy & safety testing,
promoting skill development aligned to the need of the
industry, strengthening the R&D Capability of the sector,
pricing strategy for the sector and conducive regulatory
environment.
Reference :
Press Information Bureau
Government of India
Ministry of Chemicals and Fertilizers
http://pib.nic.in/newsite/PrintRelease.aspx?relid=118080
Page
1 :
2 :
3 :
4 :
5 |
