|
Medical Plastics - New Materials & Technologies Help
Medical Device Industry
|
The Medical Device Industry is
making important contributions to advances in
healthcare supported by emerging polymeric materials
and technologies for processing of the materials.
There are numerous opportunities related to
development of products for this industry.
To deliver value to the patients,
the global medical technology industry invests heavily
in Research and Development. The level of R & D
spending in the medical device and diagnostics
industry (as a percentage of its sales) has increased
20 percent on a cumulative annual basis since 1990.
This has resulted in a significant
impact on medical technology through advances in
polymer and materials science thereby growth of the
medical polymers’ market. Medical Plastics processing
companies focus on R & D to provide innovative
polymers and high quality services to their customers.
They also aggressively collaborate and enter into
joint ventures with medical device companies.
We take this opportunity to
highlight some of these important breakthroughs by
various companies covering materials, manufacturing as
well as applications. |
Breakthroughs
• New Elastomers With 5000 % Elongation At Break
.A US based company Gelest Inc. recently introduced a new
class of elastomers that achieves unprecedented elongation
and shape-recovery properties. The silica nano particle
reinforced material approaches 5000% elongation at break;
for example, a two-yard piece of the elastomer can stretch
the length of a football field and return to its original
shape. The material can be used as the base polymer in
microfluidic devices, used for in vitro diagnostics, that
can be stretched and rolled without failure. Its
properties also hold potential for wearable medical
devices that can be stretched to suit various anatomies
and nextgeneration prosthetics.
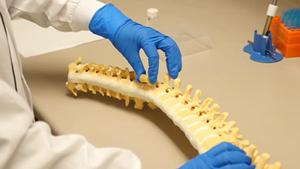
• Sponge-like Polymer Graft For Spinal Surgery
Scientists have engineered biodegradable polymer grafts
designed to repair damaged vertebrae in the spinal
column.Typically, surgeons insert expandable titanium rods
via minimally invasive surgery (MIS) to treat this
condition. Lichun Lu, PhD, of the Mayo Clinic, and her
postdoctoral fellow, Xifeng Liu, PhD, sought a more
affordable material that would work with the MIS approach.
They found a material that could be dehydrated down to a
size compatible with posterior spinal surgery, and, once
implanted, would expand to replace the missing vertebrae
through fluid absorption.
 •
Medical-Grade Polymers Revolutionize Tampon Technology •
Medical-Grade Polymers Revolutionize Tampon Technology
The Flex company USA, has developed a tampon that promises
“mess-free” sex during menstrual cycles as well as a more
comfortable overall wearing experience.
The product is a disposable, single-use menstrual product
that can be worn safely for up to 12 hours. The outer edge
of the disc is made of a proprietary blend of
medical-grade polymer, which conforms to the user’s
natural shape under exposure to body heat to create a
leak-free seal. The soft center catch collects rather than
absorbs the menstrual fluid. The circumference of the edge
is made to fit inside the vaginal fornix, covering the
cervix, and preventing menstrual fluid from entering the
vaginal canal, explains the company on its website.
• New Breast Pump Using TPE Materials
German materials firm Kraiburg TPE partnered with BeauGen
LLC to design Perfect Latch, a new product for
breast-feeding mothers. Perfect Latch acts as a cushion
between the breast pump and the mother’s skin to make it
more comfortable to pump and to protect sensitive skin
areas.
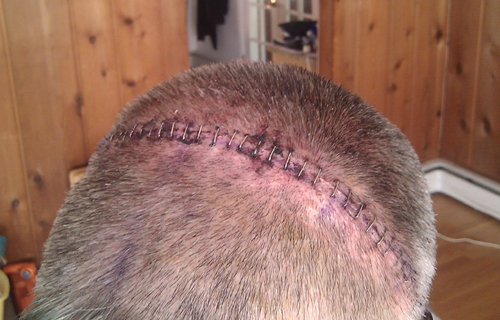 •
Innovative Materials For Bioengineered Tissue Adhesives,
Sealants And Fasteners •
Innovative Materials For Bioengineered Tissue Adhesives,
Sealants And Fasteners
Incisive Surgical Inc.’s patented Insorb 20 subcuticular
skin stapler is a sterile, single-patient-use device that
deploys up to 20 absorbable staples, sufficient to close a
17-centimeter incision. The Insorb 20 represents a new
skin closure modality designed to combine the cosmetic
result of an absorbable suture with the rapid closure
times associated with metal skin staplers.
The absorbable staple offers increased patient comfort
over metal staples while eliminating the need for metal
staple removal post-operatively. The product also provides
lower risk of infection and may shorten operative time,
compared to other wound closure methods.
The staple is composed of a copolymer of polylactic acid
and polyglycolic acid, which is absorbed by the body over
a period of a few months. The disposable stapler is made
of sterilizable acetal.
• Polyurethane Foam Pad For Wound Dressing
3M has innovated with a highly absorbent, breathable wound
dressing that is constructed from a conformable
polyurethane foam pad, and an additional absorbent
nonwoven layer from National Wovens, with a border of
transparent adhesive film impermeable to liquids, bacteria
and viruses.
The polyurethane foam pad prevents exudate pooling and
migration onto surrounding tissue while the film barrier
helps prevent external contamination and exudate
strike-through, and evaporates moisture vapor out of the
dressing. The 3M foam adhesive dressing is used for
treating moderate to highly exuding dermal wounds such as
pressure ulcers, neuropathic ulcers, abrasions and first-
and second-degree burns.
Page
1 |
