| DUR/ODR
The method of describing the uniformity of dose in
a given product is the concept of Dose Uniformity
Ratio (DUR), which is the ratio of the maximum
dose in a product container to the minimum dose in
the container. This ratio increases with increase
in the density of the product as well as with the
size of the container/tote box
Validation of radiation
processing
• IQ – INSTALLATION
QUALIFICATION
• OQ – OPERATIONAL QUALIFICATION
• PQ – PERFORMANCE QUALIFICATION
Installation Qualification
Installation qualification is
the process of obtaining and documenting evidence
that equipment (the irradiator and its associated
processing equipment and measurement instruments)
has been provided and installed in accordance with
its specification
Operational Qualification
Operational qualification is
the process of obtaining and documenting evidence
that the installed equipment operates within
specified limits when used in accordance with its
operational procedures. It is carried out after
the commissioning of the facility.
Performance Qualification
Performance qualification is
the process of obtaining and documenting evidence
that the equipment, as installed and operated in
accordance with operational procedures,
consistently performs in accordance with specified
criteria and thereby yields product meeting its
specification
Dosimetry Systems used for
Radiation dose measurement
|
Dosimeter |
Measurement instrument |
Dose
range (kGy) |
Remarks |
| Alanine |
EPR
spectrometer |
0.001-100 |
Various
shapes: rods, pellets |
| Dyed
PMMA |
Visible
spectrophotometer |
1-100 |
1 × 3
cm 3 mm thick |
| Ceric-cereous
sulphate solution |
Potentiometer, UV spectrophotometer |
1-100 |
Glass
ampoules Diameter~12 mm |
| Fricke |
UV –Vis
spectrophotometer |
0.04 –
0.4 |
PP
vials |
|
Dichromate |
UV –Vis
spectrophotometer |
1-100 |
Glass
ampoules Diameter~12 mm |
Regulatory Bodies/Standards
for gamma Sterilization facilities
Radiation sterilization process
is currently regulated by two standards, ISO 11137
and ISO13485
• In India apart from
these standards CDSCO/FDA is regulating the
radiation sterilization process for health care
products according to MDR 2017
• AERB (Atomic Energy Regulatory Body)-India
• Medical Devices Risk Analysis – ISO 14971
• ISO 9001 (QMS)
Quality Management
System
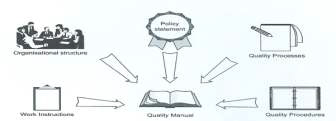 |
RECENT DEVELOPMENTS IN
SAFETY FEATURES
What are current standards for
quality/c GMP practised by Irradiators worldwide ?
• MHRA/UK
• USFDA
• TGA –Australia
• UNFPA
•
WHO/Drug and Cosmetic Act , India MDR 2017 / ISO
13485 / ISO 14971
• ANVISA-BRAZIL
• MCC- South Africa
• PMDA- Japan
• NMRA-Srilanka
• MDA - Malaysia
• ANSM-France
• CDSCO –India
CURRENT STATUS OF RADIATION STERILISATION
TECHNOLOGY
• India largest exporter of IUDs to UNFPA
• Medical Devices Act 2016 (Impending legislation)
• Synergistic growth in medical devices sector
• Approximately 0.175 Million Cu. M. medical
products sterilized by Gamma currently
• With 23 Radiation Processing Facilities
operating in the country currently FUTURE IS
PROMISING !!
Current trend in radiation technology in
India
• Captive gamma radiation facility
• Contract gamma radiation facility
• Capacity of each facility 1.5Mci to 5MCi
• Total gamma radiation facility in India – 38
• Total Co-60 radiation source – 38Mci as on 2023
• 10MeV 10kW Electron beam facility terminal
sterilization is also coming up.
Role of BRIT-DAE
• Developmental initiatives with changing
trends in quality, safety and security of
industrial irradiators
• Recently developed FOAK (First Of A Kind) -
animated training simulator named ISOTRAIN for
Radiation Processing Facilities
• BRIT also launched ISOCAD (Integrated System of
Computer Aided Dosimetry system) – A c GMP
Compliant CCPDMS
Gamma Lrradiators Worldwide
There are now about 200 commercial Co–60
irradiators for radiation sterilization, operating
in 47 countries worldwide, containing
approximately 240–260 MCi of gamma emitting Co- 60
Sources
Merits of gamma Sterilization
• Simple Process Only parameter to control is
exposure time
• Pre-packed in hermetically sealed packages,
impermeable to microorganisms, before and after
sterilization.
• Temperature is not raised much: compatible with
temperaturesensitive materials, such as
pharmaceuticals and biological samples
• Flexibility: can sterilize products of any
material, size, or thickness
• Leaves no chemical residue
• Immediate Availability of product after
sterilization
• Gamma radiation easily reaches all parts of the
object to be sterilized due to its high
penetrating ability
Limitations of gamma Sterilization
• Material Degradation : few material degrade
after terminal sterilization radiation dose
(brittle, colour change)
• Method is costly alternative to heat
sterilization |
